|
(c) Similarities: Both are orbitals that can contain two electrons. For a molecule, ψ represents a mathematical combination of atomic orbitals. Differences: ψ for an atomic orbital describes the behavior of only one electron at a time based on the atom. Hcn lewis structure molecular geometry hybridization polar or nonpolar 1. The valence electrons on the Hydrogen atom and lone pairs present repel each other as much as possible to give. From the Lewis structure, it can be observed that there are two symmetrical NH2 chains. The natural first combination is a 50 50 50 50 split to form two sp s p orbitals, one directed to the H H and the other directed toward. (b) Similarities: Both are quantum-mechanical constructs that represent the probability of finding the electron about the atom or the molecule. As we discussed earlier, the Lewis structure of a compound gives insight into its molecular geometry and shape. Hybridization of HCN can be calculated using the following formula. In HCN H C N, we hybridize/combine the two remaining orbitals on the carbon atom to form two bonding orbitals, one to the hydrogen, another to the atom on the other side of the carbon (a C C or an N N ). Because the methane molecule has four electron domains (four hydrogen atoms. Because hydrogen and nitrogen tend to be far apart, HCN takes on a linear structure. What is the electron geometry, molecular geometry, and hybridization of HgBr2. The linear molecular geometry of hydrogen cyanide has bond angles of 180 degrees. Therefore, both the electron and molecular geometries are linear. As a result, the hybridization for the HCN molecule is an sp hybridization. The steric number is 2, and there are no lone pairs on the central atom. Differences: σ orbitals are end-to-end combinations of atomic orbitals, whereas π orbitals are formed by side-by-side overlap of orbitals. The number of sigma bonds created equals the number of hybrid orbitals. (a) Similarities: Both are bonding orbitals that can contain a maximum of two electrons. 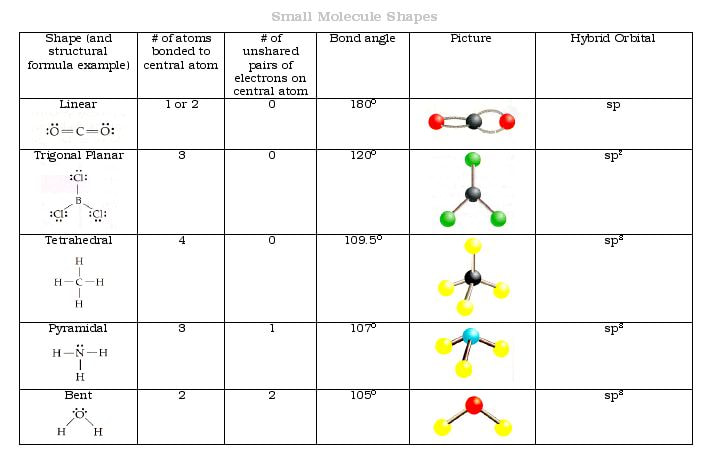
(d) What hybridization is consistent with the shape you predicted? (c) Predict the shape of the molecules of the compound. (b) Write a Lewis structure for the compound. Analysis of the compound indicates that it contains 77.55% Xe and 22.45% F by mass. A mixture of xenon and fluorine gases, confined in a quartz bulb and placed on a windowsill, is found to slowly produce a white solid. (c) Determine the hybridization of each type of carbon atom.įor many years after they were discovered, it was believed that the noble gases could not form compounds. 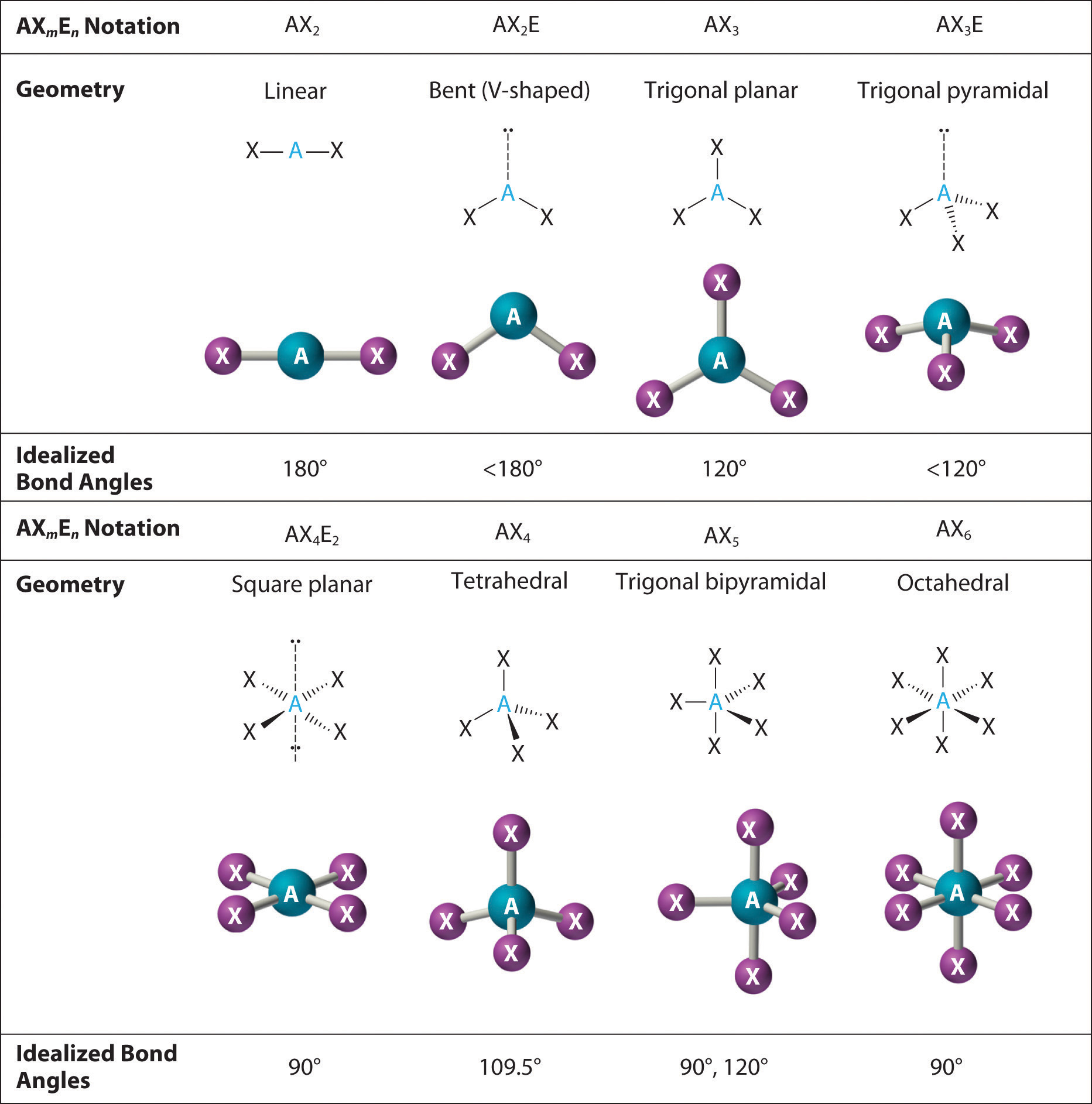 
(b) Predict the geometry about the carbon atom. As this molecule has a linear molecular geometry, HCN has bond angles of 180. (g)\)įor each of the four carbon compounds, do the following: structure, molecular geometry, polar or non-polar, hybridization.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
